10.4: The Ideal Gas Equation - Chemistry LibreTexts

By A Mystery Man Writer
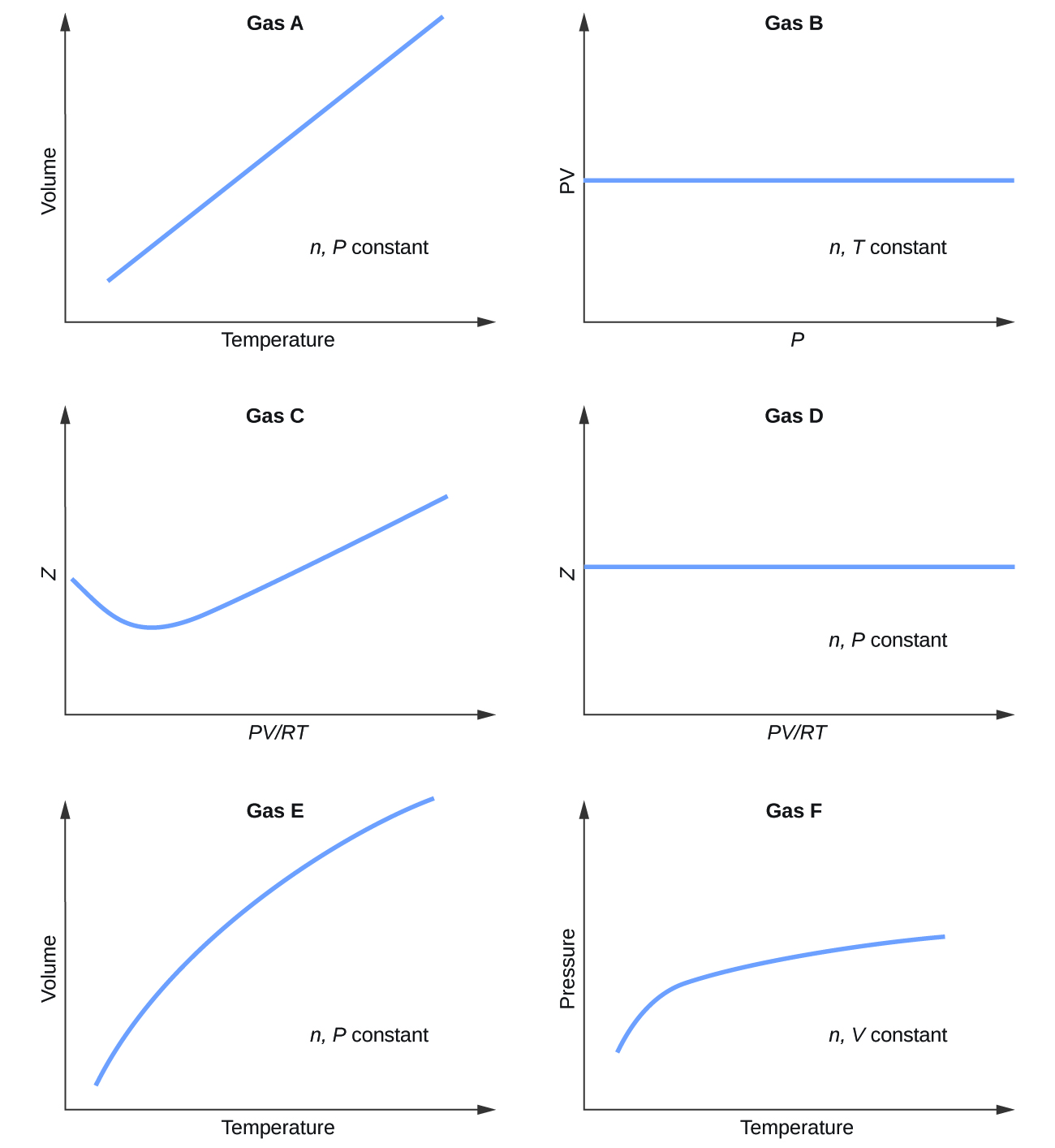
The empirical relationships among the volume, the temperature, the pressure, and the amount of a gas can be combined into the ideal gas law, PV = nRT. The proportionality constant, R, is called the …
The empirical relationships among the volume, the temperature, the pressure, and the amount of a gas can be combined into the ideal gas law, PV = nRT. The proportionality constant, R, is called the gas constant. The ideal gas law describes the behavior of an ideal gas, a hypothetical substance whose behavior can be explained quantitatively by the ideal gas law and the kinetic molecular theory of gases. Standard temperature and pressure (STP) is 0°C and 1 atm.

Physical Chemistry PDF, PDF, Fahrenheit

Gas Law Practice Problems All Types of Gas Law Examples

Equation of State, Glenn Research Center

are you an action potential bc youre all i think about💔 The energy n, electrolysis in chemistry

3.4 The Wavelength Nature of Matter – Chemistry LibreTexts – Chemistry Fundamentals

Foods, Free Full-Text
Ideal Gas Law - Wyzant Lessons

The Ideal Gas Law - Chemistry LibreTexts, PDF, Gases
The Ideal Gas Law - Chemistry LibreTexts, PDF, Gases

The Ideal Gas Law - Chemistry LibreTexts, PDF, Gases

Physical Chemistry PDF, PDF, Fahrenheit