Sunday, Sept 29 2024
Does atomic size increase down a group?

By A Mystery Man Writer
Does atomic size increase down a group?
Does atomic size increase down a group

ReasonAtomic size depends upon the valence shell electronic configuration.AssertionThe atomic size generally increases across a period and decreases down the group.

Trends in the periodic table - Labster

Periodic classification of elements #26

AP Chapter 7 – Periodic Properties of the Elements - ppt download

6answer Module Sk015 Chapter 3 1 .pdf - Practice Module Chapter 3
Define atomic size. How does it vary in a period and in a group?

File

Ionisation Energy Teaching Resources

Chapter 6: Periodic Trends. - ppt download
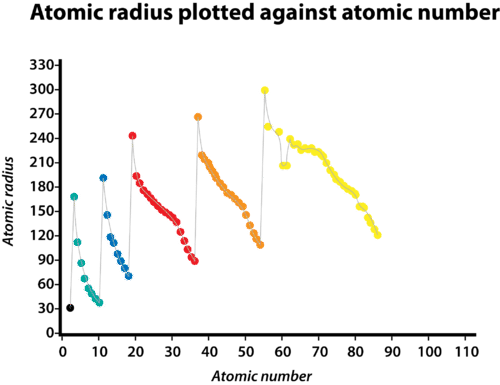
Periodic Trends in Atomic Radius

Periodic Trends 9: Why Atomic Radii Increases Down a Group

Silence of the Leukemic Clone
Related searches
Related searches
- 70S Series 7 Replacement Head for Braun Electric Foil Shaver, Compatible with Braun Series 7 790cc 760cc 750cc 720 799 797 - AliExpress

- Abercrombie & Fitch Long Sleeve Ponte Corset Bodysuit

- Porsche Mission R: A taste of motorsport's bold electric future

- Ribbed sweatpants

- BROOKS, Barry Loy - Obituary - North Bay - North Bay News

©2016-2024, slotxogame24hr.com, Inc. or its affiliates