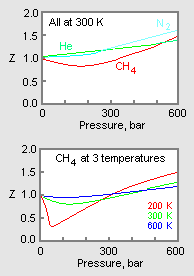
Compressibility factor Z = PV / nRT is plotted against pressure as

By A Mystery Man Writer
Compressibility factor Z = PV / nRT is plotted against pressure as shown below:What is the correct order for the liquefiability of the gases shown in the above graph? A. CO 2
Compressibility factor Z - PV - nRT is plotted against pressure as shown below-What is the correct order for the liquefiability of the gases shown in the above graph- A- CO 2- CH 4- N 2- H 2B- H 2- CH 4- N 2- CO 2C- CH 4- H 2- N 2- CO 2D- H 2- N 2- CH 4- CO 2

Chapter 5 Gases
Untitled Document
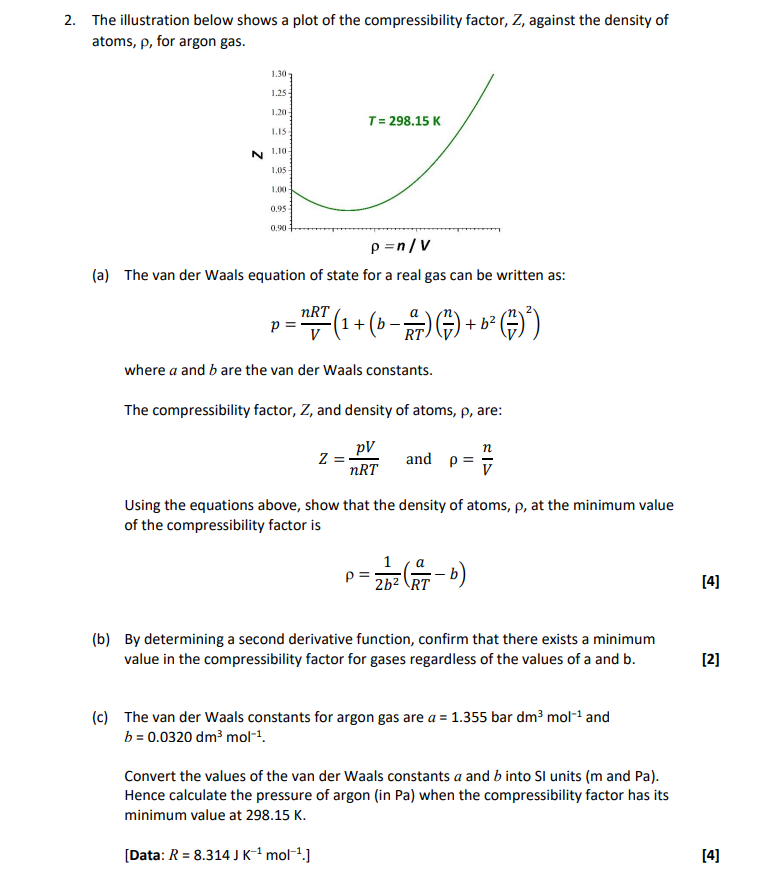
Solved 2. The illustration below shows a plot of the
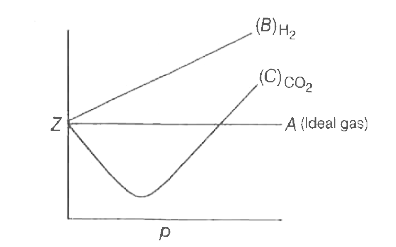
Telugu] The variation of compressibility factor (Z) with pressure (p
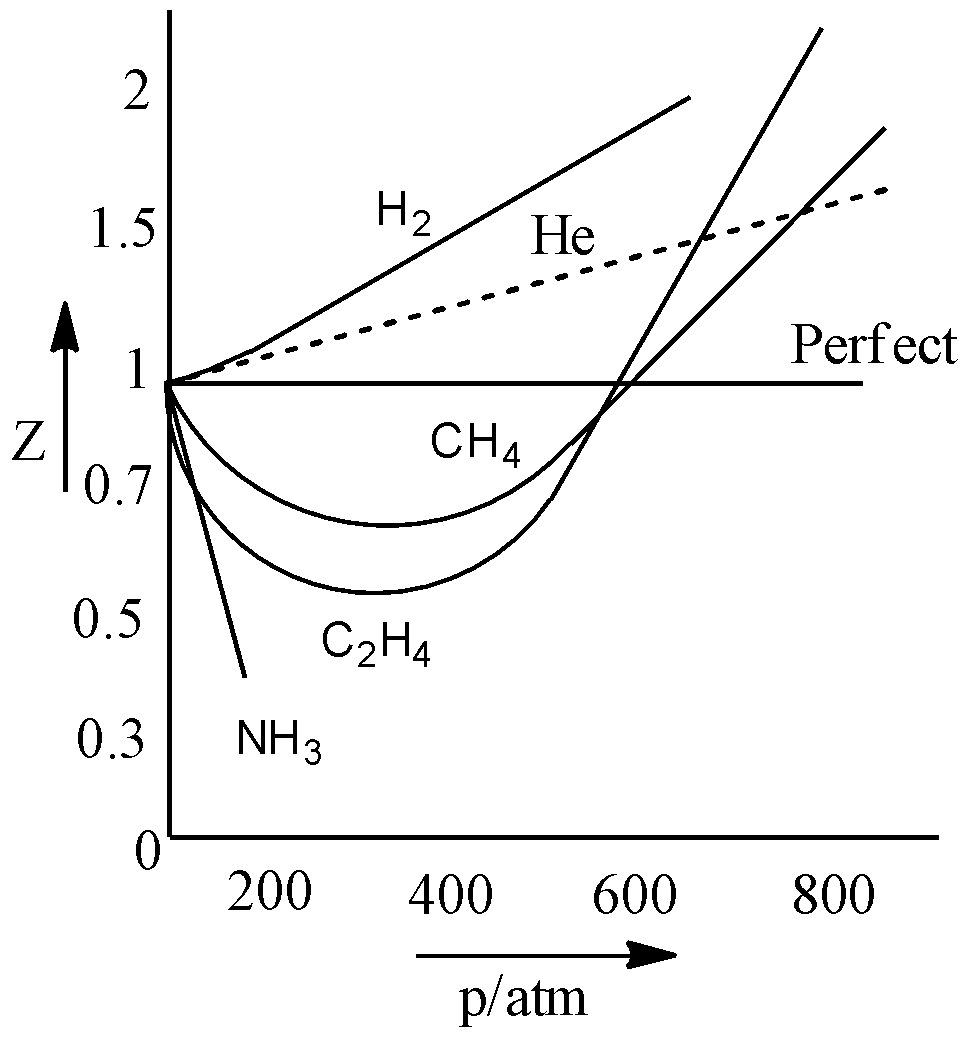
The answer to this question is a single digit integer, ranging from 0 to 9.The compressibility factor Z for an ideal gas will be________.
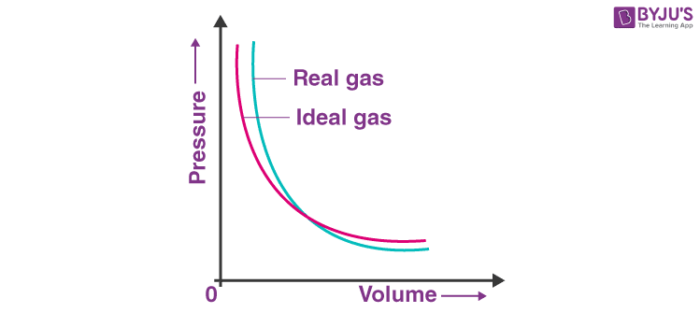
Deviation Of Real Gas From Ideal Gas Behavior

Explain the shape of graph obtained between pressure P and 1/v for perfect gas at constant temperature? - Quora

Compressibility Factor Calculator - File Exchange - MATLAB Central

Real gases

A real gas M behaves almost like an ideal gas. Graph 1 is obtained by plotting volume, V against temperature, T for x mol of gas M at pressure, P_1. a. Suggest