Solved 1. A cup of hot 50°C water is poured onto 35g of ice

By A Mystery Man Writer
Answer to Solved 1. A cup of hot 50°C water is poured onto 35g of ice

Mogo - HOTTIPS MICRO USB 4FT

SOLVED: A piece of ice of mass 40 g is added to 200 g of water at 50oC. Calculate the final temperature of water when all the ice has melted. Specific heat

single serve dessert with 19 grams of quality protein 👇👇 🍫I put my

Culinaire #2:7 (december 2013 by Culinaire Magazine - Issuu

An ice cube whose mass is 50 g is taken from a refrigerator where its temperature was`-10^C`. If
/@api/deki/files/57428/CNX_Chem

Calaméo - Guide d'achat 2021-2022 EN
How many cups of coffee are equal to one shot of espresso? - Quora
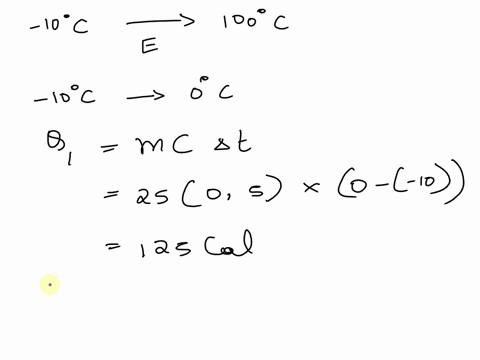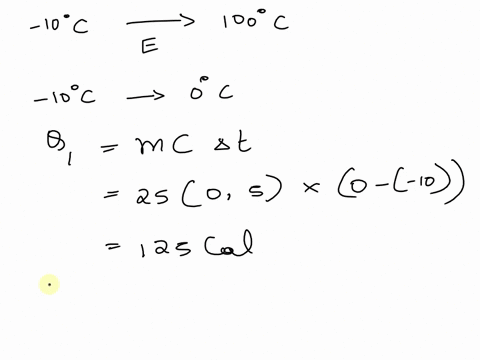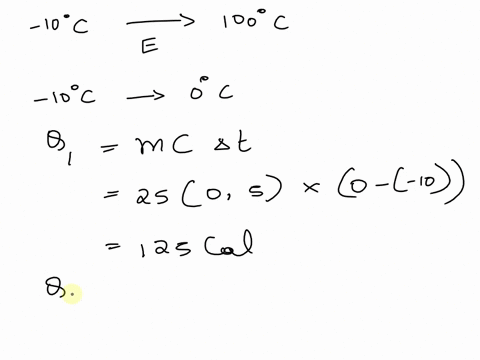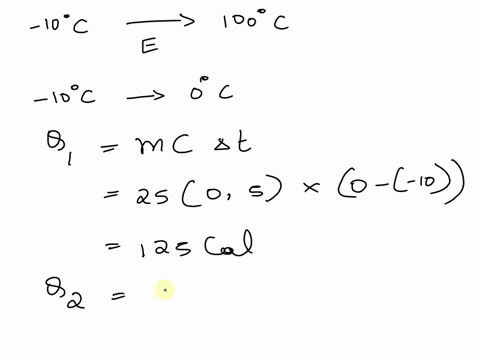
SOLVED: A cup of hot 50°C water is poured onto 35g of ice at 0°C until the entire system ends up at 20°C. How much energy is needed to melt the 35g

Mogo - San Pellegrino Sparkling Water 16.9Oz
Solved A.) to heat 20.6 g of water from 12.5∘C to 30.0
- SOLVED: A 35-g ice cube at 0.0 °C is added to 110 g of water in a 62-g aluminum cup. The cup and the water have an initial temperature of 23 °C. (
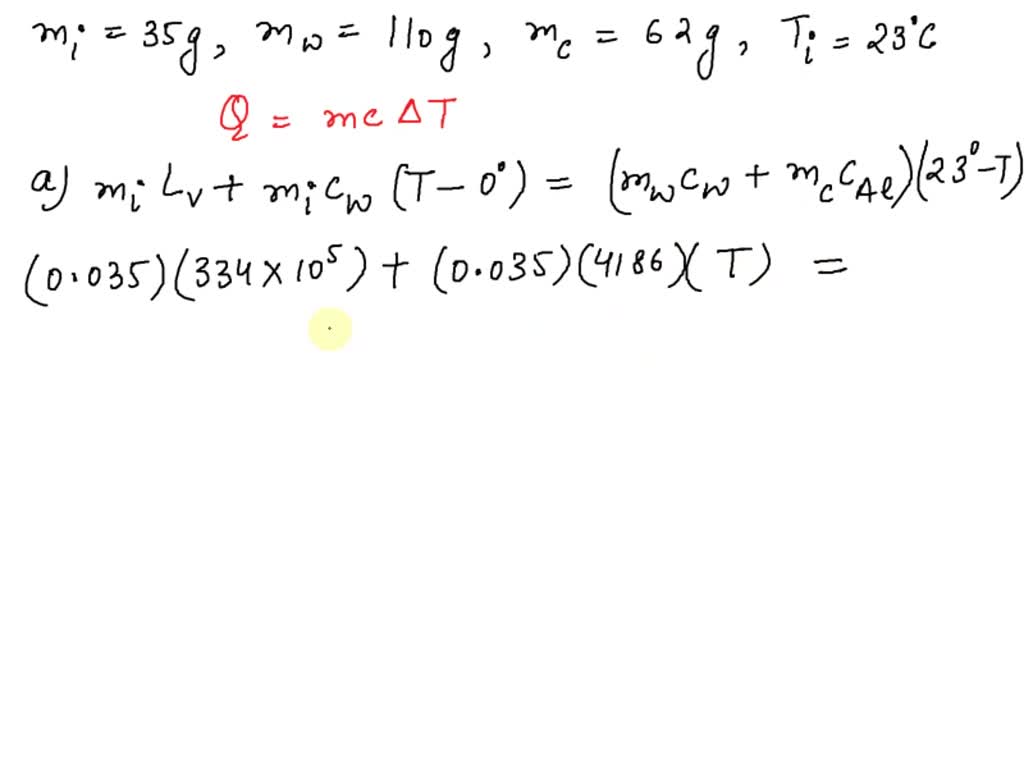
- Weight Conversion Chart for Baking, recipe, baking, weight, Don't let recipe conversions keep you from baking something. Use my Weight Conversion Chart for Baking! Get a FREE Downloadable Chart here
- OffLimits, Spark Strawberry Cereal – Basquet

- Continental Soup Cup-A-Soup Snack or Light Meal Cup Ct Cream Chicken Soup Corn Bigger Single Serve, 35 g : : Pantry Food & Drinks

- Buy Knorr Instant Porridge Chicken Cup

- Men's Underwear - The Factory Outlet

- KIHOUT Pants For Women Deals Women's Summer High Waisted Solid Color Capris Matching Slim Fitting Yoga Gym Pants

- Candyman 99592 Harness-thongs Outfit Black

- Underwear Panties Panty Shaper Sissy Transgender Underpant XL/2XL Briefs

- Plus Size Work Dresses - Shop Women's Work Wear

